 In this case, the products, or final state, have more free energy than the reactants, or initial state. Spontaneous is equivalent to an exergonic reaction whereas non- spontaneous is equivalent to an endergonic reaction.Įxergonic reactions by definition are spontaneous processes whereas endergonic reactions, also by definition, are non-spontaneous processes. There are two types of processes (or reactions): spontaneous and non-spontaneous. Reactions with a positive G (G > 0), on the other hand, require an input of energy and are called endergonic reactions. By net, the implication is that one is considering more than just activation energy, that is, not simply the energy required to get a reaction started. By contrast, if something proceeds not spontaneously then it requires a net input of energy. Spontaneous reactions occur without any input into the reaction, non-spontaneous reactions need input such as extra energy. The spontaneous production of mediators in these clonal mast cell disorders. 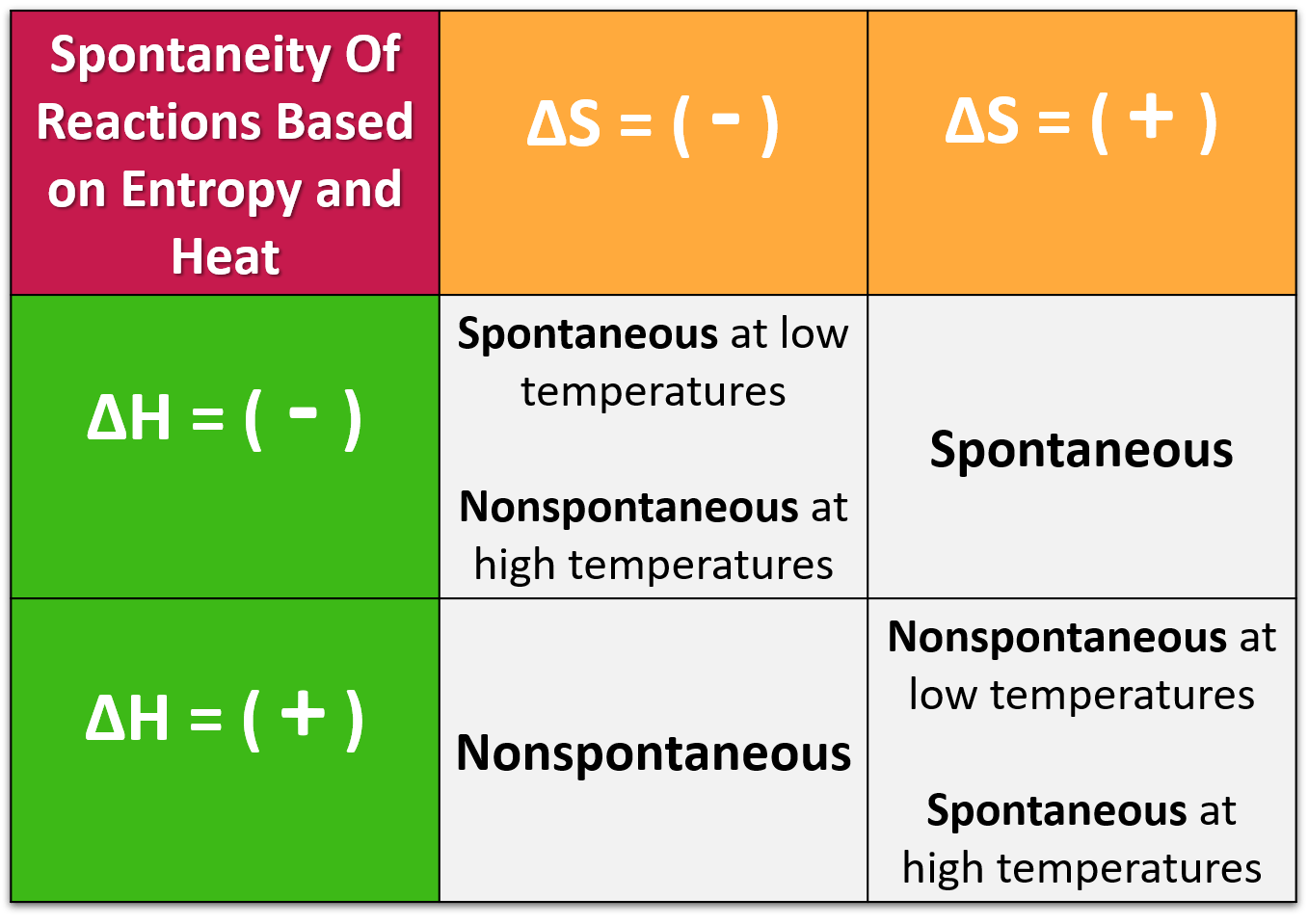
In order for a reaction to be nonspontaneous, it must be endothermic, accompanied by a decrease in entropy, or both. Mast cells are allergy cells responsible for immediate allergic reactions. Note that movement down a concentration gradient or, more generally, towards equilibrium or simply exergonic reactions, are spontaneous processes.įigure legend: Spontaneous processes overall result in a reduction in energy associated with the process. A nonspontaneous reaction is a reaction that does not favor the formation of products at the given set of conditions. In biological systems it is typically those chemical reactions that do no require an input of ATP, or equivalent energy source, that are spontaneous reactions. this video will discuss how non spontaneous reactions are able to go. That is, if output is greater than input, in terms of a quantity known as free energy, then the process is described as spontaneous. A spontaneous reaction is a reaction that favors the formation of products at the conditions under which the reaction is occurring. Instead, it is whether the process, once it proceeds, particularly post any transition state, ends up liberating more energy than was put into the system to get the process started. Spontaneous Reaction and Gibbs Free Vitality 1-4 What Causes a Reaction to exist Spontaneous.  
That input of energy, however, is not the determinant of spontaneity. Processes in general require an input of energy to be initiated, called activation energy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
